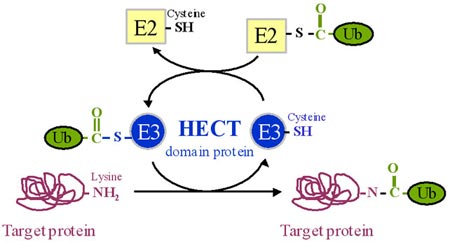
A HECT domain was first discovered in the C-terminus of the E6 associated protein (E6-AP) which targets p53 for rapid degradation by the ubiquitin-proteasome system in the presence of the human papilloma virus oncoprotein E6. Lateron homologous domains have been found in serveral other proteins involved in specific protein degradation by the ubiquitin-proteasome system and have therefore been termed Homologous to E6 -AP Carboxyl Terminus (HECT). HECT domains consist of ca. 350 amino acids and contain a conserved cysteine residue which is necessary for the tagging of target proteins with ubiquitin.
The varying N-termini among the different HECT-domain proteins are probably involved in specific recognition of proteins that have to be degraded. Examples of HECT domain proteins and their target proteins:
|